 If ice were denser than the liquid, the ice formed at the surface in cold weather would sink as fast as it formed. In fact, the ice forms a protective surface layer that insulates the rest of the water, allowing fish and other organisms to survive in the lower levels of a frozen lake or sea. Because ice is less dense than liquid water, rivers, lakes, and oceans freeze from the top down. The structure of liquid water is very similar, but in the liquid, the hydrogen bonds are continually broken and formed because of rapid molecular motion. The electrochemical desulfurization in the molten salt is a promising green route to extract metals directly from sulfide ores.However, less attention has been paid to the process of converting solid sulfides to liquid metals, especially the evolution of the solid sulfide/liquid metal interface. A liquid is a state of matter in which atoms or molecules are constantly in contact but have enough energy to keep changing positions relative to one another. By adding heat energy, a substance can change from a solid to a liquid. Here are three-dimensional views of a typical local structure of water (left) and ice (right.) Notice the greater openness of the ice structure which is necessary to ensure the strongest degree of hydrogen bonding in a uniform, extended crystal lattice. Materials can be changed from one state to another by heating or cooling (freezing). This expansion is minimum in solid compared with liquid and. To change the state of a substance energy must. When heat is added, the molecules gain energy and vibrate and force other molecules to move apart. 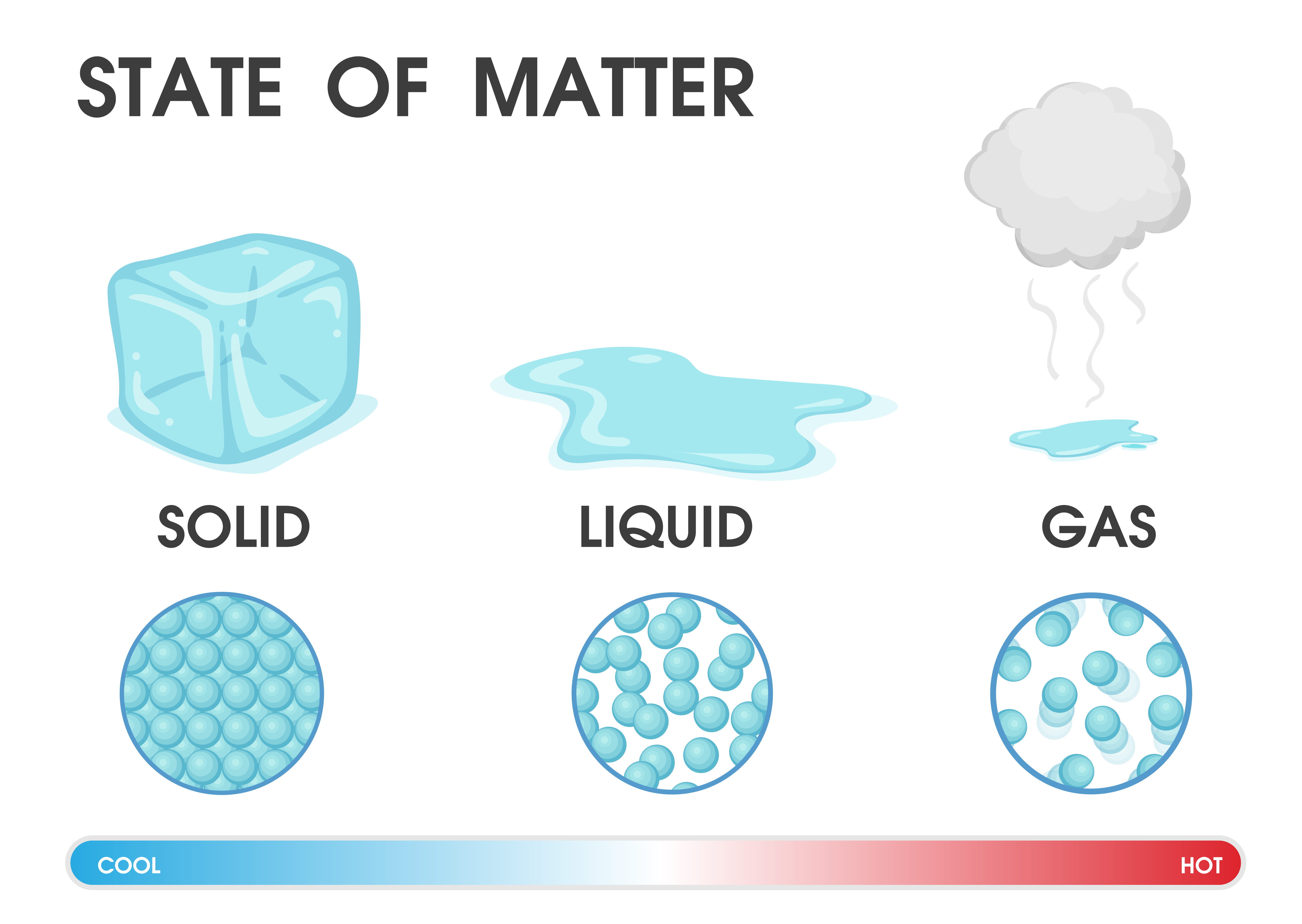
\): Three-dimensional views of a typical local structure of liquid water (left) and ice (right). Physics Solids, liquids and gases Key points Substances can exist in three states of matter - solid, liquid and gas. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
